Honey is one of the most frequently adulterated food products in the world. At first glance, it may appear flawless – golden, fragrant, and enticing – yet it is often diluted with inexpensive sugar syrups derived from sugar beet or corn. Such adulteration is nearly impossible to detect by sight or taste alone, which is where science steps in.
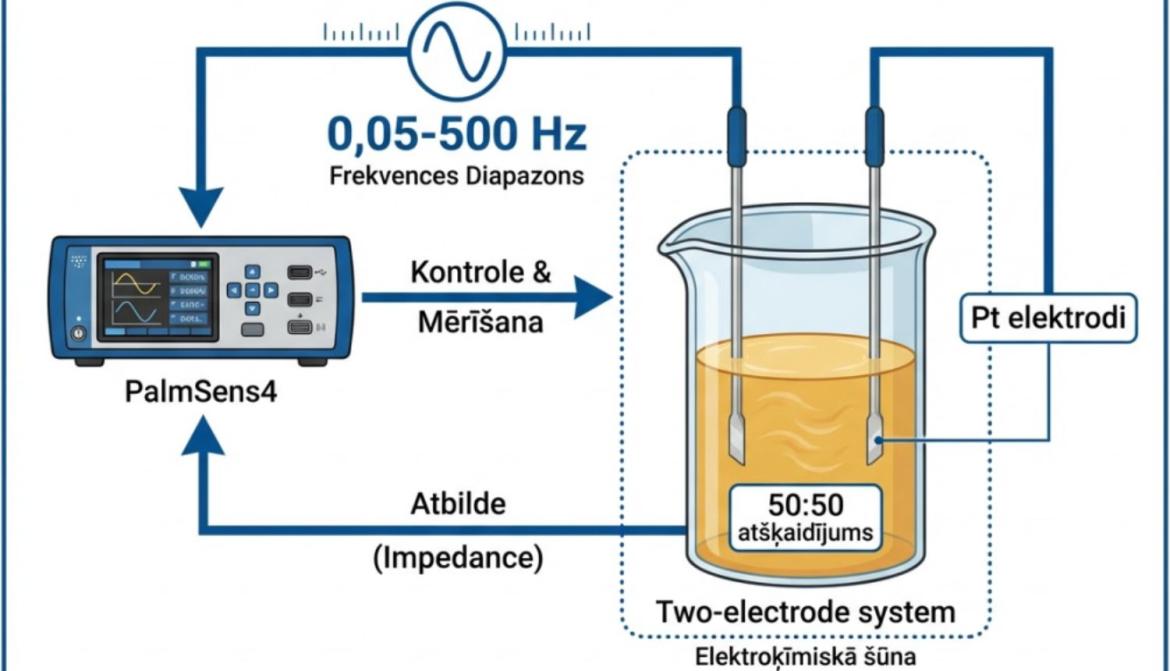
A research team from the University of Latvia Faculty of Medicine and Life Sciences, Department of Chemistry – Dr. Vitālijs Lazarenko, Prof. Dr. chem. Arturs Vīksna, Mg. chem. Māris Bērtiņš, Mg. chem. Krišs Dāvids Labsvārds and Vladlens Laškovs is developing a rapid and efficient approach to verifying honey authenticity using a technique with a complex name: electrochemical impedance spectroscopy (EIS). In simple terms, this method measures how honey responds to a very weak electrical signal.
Why does this approach work? Honey is far more than just sugar – it contains organic acids, minerals, enzymes, and other biologically active compounds. These substances influence its electrical conductivity. When sugar syrup is added, the chemical composition changes, and so do its electrical properties.
During the measurement process, two platinum electrodes are inserted into a honey sample, and a very weak alternating current is applied across a range of frequencies. The device records impedance, or complex resistance. The resulting data are then analysed using mathematical models to determine specific parameters.
The study identified two key indicators: solution resistance and charge transfer resistance. Researchers found that as the proportion of added syrup increases, solution resistance rises significantly, enabling the detection of adulteration. Meanwhile, charge transfer resistance varies among different types of honey, opening up the possibility of determining geographical or botanical origin in the future.
The entire analytical process takes only a few minutes and requires no complex sample preparation. However, several additional steps are needed before the method can be widely implemented in routine honey authentication. First, a comprehensive database of honey samples with electrochemical profiles must be established to enable rapid and reliable comparisons – not only to detect adulteration but also to determine origin. Second, the device itself requires optimisation, for example by replacing platinum electrodes with a laboratory-developed sensor that could enhance sensitivity while reducing costs.
If these improvements are successfully implemented, the method could become a competitive and widely adopted solution for detecting honey adulteration.
The research was conducted at the Faculty of Medicine and Life Sciences of the University of Latvia within the project "Electrochemistry for the Future: Advancing Education and Science." It was funded through the University of Latvia Foundation project competition "MikroTik Grant Competition in Natural, Technological and Medical Sciences."


